Physicist: The short answer is; you probably can, but not for long.
If you’ve taken a little chemistry you probably know that the electrons in an atom “stack up” in energy levels. The more electrons you add, the higher the electrons have to stack. The same is true for the protons and neutrons inside the nucleus of the atom. What’s a little surprising is that the stack for the protons and the stack for the neutrons are independent of each other.
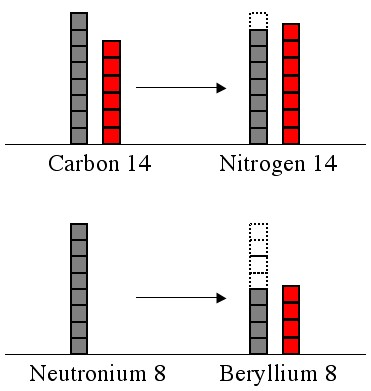
In a stable atom the energy in the proton stack will be about the same as the energy in the neutron stack. If they’re unequal, then a neutron will turn into a proton ( decay), or a proton will turn into a neutron (
decay) to even out the levels. The greater the difference the sooner the decay, and the more radioactive the atom. There are plenty of exceptions (e.g., Uranium 237 vs. 238), but the pattern usually holds.
An atomic nucleus made entirely out of neutrons (known to some sci-fi aficionados as “neutronium”) would be completely imbalanced and would decay instantly. It would be tremendously radioactive.
Chemistry nerds may have noticed that heavier elements have more neutrons than protons. For example, Iron 58 has 26 protons, 32 neutrons, and is stable. Forcing protons together takes a lot of energy (likes repel), so after hydrogen, proton energy levels get higher, quicker, than than neutron energy levels.
Exception! In neutron stars you have the added component of lots of gravity. When a proton and an electron fuse into a neutron they take up less room, and since gravity wants to crush everything together, this is a lower energy state. So neutron stars are the one example of stable neutronium. However, most people would say that calling an ex-star an atomic nucleus is pushing the definition of “atomic” a bit far. Even more exciting; neutron stars may also contain the only naturally occurring stable lambda particles!
The picture of the completely ordinary chemist is from here.








Pingback: Q: What are Feynman diagrams, how are they used (theoretically&practically), and are there alternative/competing diagrams to Feynman’s? « Ask a Mathematician / Ask a Physicist
Pingback: Q: What is radioactivity and why is it sometimes dangerous? | Ask a Mathematician / Ask a Physicist