Physicist: The long answer is really, really long. Superconductivity comes in a variety of different forms, and there’s a different explanation for each. To sum them up in a thumbnail sketch of a thumbnail sketch:
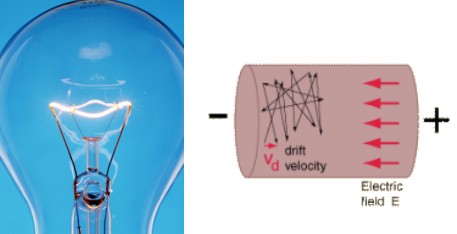
In conductors, the primary cause of electrical resistance is the exchange of kinetic energy between the moving electrons and the material they’re moving through. As a result, the material heats up and the electrons find themselves knocked in random directions so much that you can barely tell what direction they’re supposed to be moving in. In fact, the net movement of an electron in the direction of the current is often referred to as “drift velocity”.
By making the material cold there is less energy to knock the electrons around, so their path can be more direct, and they experience less resistance. So far this is just an argument for why coldness causes lower resistance, but says nothing about superconductance. Enter quantum mechanics. As the name implies, quantum mechanics describes how everything in the universe is quantized (not continuous). The energy that gets exchanged must be in discrete (quantized) chunks that have a minimum size. If the material is cold enough, then there isn’t enough heat energy available to reach that minimum “chunk”. So energy is not passed from the material to the electrons, the electrons don’t get knocked around, and they don’t experience any resistance.
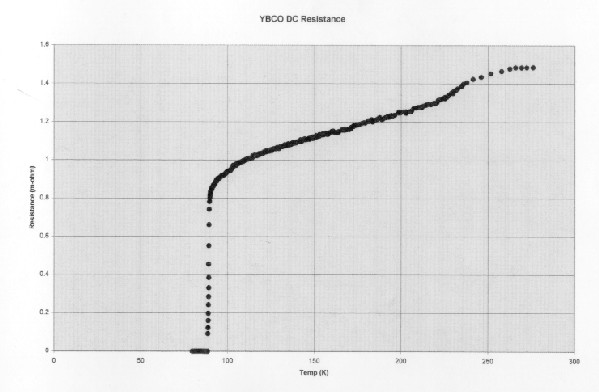
Temperature vs. Resistance in Yitrium-Barium-Copper-Oxide, the first "high temperature" superconductor. This was a breakthrough because YBCO can be cooled to superconductive using liquid nitrogen, which is cheap.
It turns out that many materials become a superconductor if cooled down enough. In fact, superconductivity was discovered accidentally when a sample of Mercury was cooled to 4° K (-452° F or -269° C). The slick new superconductors also take advantage of the “can’t exchange a chunk that big” technique, but involve lots of QM tricks to force the operational temperature way up. Figuring out these tricks and putting them together correctly is what makes new higher temperature superconductors so difficult to invent.
It’s worth noting, finally, that the answer above is the most intuitive answer I can come up with, and not the most correct. A more precise understanding involves a more thorough understanding of the math and wave mechanics (the electrons act more like waves than particles, and the material acts more like a waveguide than a hose).







Is it possible that atoms could ever behave like sub-atomic particles in that they appear to be able to be in two different places at the same time?
Hells yes!
There’s a paper here about the successful measuring of Carbon 60 (Bubkyballs) in multiple places. It really just a rehashing of the double slit experiment.
Thanks. That was a nice explanation for an interested layman like me. So, Does that mean that most wires would become better conductors in space?
Pingback: Vino tinto: el secreto de la superconductividad
I take all of this to mean. It depends on which type of superconductors are being used.
So the correct answer is a variable. That explains the innate properties of the materials. Combined or multiplied with the force being applied.
I think personally. It’s better explained as a force molecularly amplified by an reflective effect between certain differentially aligned materials. That may become atomically aligned at different temperatures. A feed back loop or self supporting magnetic phenomenon. Perhaps even simplified as the explanation of atomic shockwaves between a certain distance of molecules.
Although I’m not sure if any of these explanations or hypothesis could adequately describe the science. In one sentence or paragraph. Probably because there are coincidences between field particle and wave physics. Where electricity is described as a conductive force, Just as physical shock or magnetic lock levitation. These differences all roll into the superconducting category.
If the explanation is trying to show the amplified or reflective force of a superconductor.
It can be difficult to explain in a sentence or mathematical expression. Without specifics on the materials and forces involved.
However there is still a correlation between the science. Superconductors can change in atomic structure with the extreme cold. I think this has to do with the physics of the atomic polarity within the material or elements. Which forms a natural amplifying force relative to gravity or pre solar fission, magnetism and radiation. Which could be the result of residual interstellar effects. Cosmic radiation and spacial debris collisions. The compression of wave effects. Into a more focused ripple effect.
Perhaps this level of physics. Deals with the observation of atomic monopole flipping phenomenon. As a result of the extreme cold. But it could just as easily be the physical movement of the atomic structure by an external force. Like pushing the electrons faster then the conductive materials will allow transduction.