The original question was: Is it possible for something to “feel” colder than absolute zero? If the forecast called for 1K (1 Kelvin) with 20mph gusts of wind, would the wind chill be below 0K?
Physicist: This is a beautiful question!
“Wind chill” is a very ad hoc, artificial measure used to describe the fact that cold air pulls heat out of us faster when it’s moving.
When air is stagnant and you’re standing still, you’ll be surrounded by a bubble of warm air (heated by body heat). This bubble insulates us from the surrounding air temperature. So, if it’s 40°F out, and there’s no wind, you’ll actually be losing heat as though it were warmer, because near your body it is warmer. In fact, this is the whole point of clothes (beyond the whole modesty thing): keeping a layer of air near the body. The insulation provided by the cloth is generally dwarfed by the insulation provided by the air it holds in.
So, when you walk outside and say “it feels like it’s about 70°F out” what you really mean is “the bubble of air around me feels like it’s about 85°F, and my experience tells me that corresponds to an ambient temperature of 70°F”. But you’d need to be kind of a jerk to go through all that. Instead you just say “feels like 70°F” (even though what you’re experiencing is actually warmer).
When the wind is blowing however, that layer of warm air is pushed away and we’re exposed to the actual air temperature. What we call the wind chill temperature is the temperature it would have to be in order for you to lose body heat at the same rate, were the air to be sitting still.
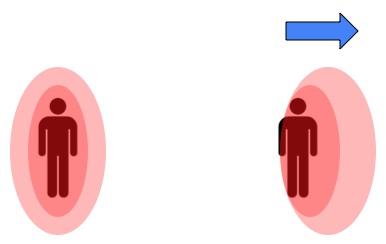
Being not just squishy but warm as well, we heat up the air immediately around our bodies. This air insulates us from direct exposure to the surrounding cold air. When there's wind that bubble gets thinner and with less insulation we lose heat faster. This picture is not to scale, and is a poor representation to boot.
Wind chill is pretty subjective (people of different sizes and shapes will experience wind chill differently), and not terribly exact, but the standard gives you a decent ball park estimate.
If you were to stick your hand into liquid helium at 1K, you would again get a layer of slightly warmer liquid helium around your hand (I’m using liquid helium here because it stays liquid essentially all the way to 0). If you were to stick your hand in flowing liquid helium, that warmer layer would be washed away and your hand would be exposed to actual 1K helium.
As strange as it sounds, it is perfectly fair to say that, with “wind chill”, the temperature is below 0K, because that’s how cold stagnant helium would have to be to match the heat loss caused by 1K helium that’s moving.
There’s nothing wrong with this statement. Heat loss is governed by the difference in temperature between a body and its environment. Regardless of whether or not one of the temperatures involved is below zero.







Surely the answer is simply ‘No’!
The clue is in the name. At absolute zero all molecular movement ceases. As you say ‘wind chill’ is the process where the small insulating gradient between your skin and the true environmental temperature is stripped away. If we assume the helium flow can do this then at the instant your skin feels the ‘wind chill’ then it would be feeling the true temperature. Being absolute zero then all molecular movement would cease and nerve signals would be unable to transmit the ‘feeling’. What you would feel would be the temperature at some subcutaneous level which is still above absolute. Therefore it is not possible to feel temperatures below absolute.
I agree with John?
I’m pretty sure you wouldn’t feel anything, whether the Helium was flowing or not.
@John
Well, the question does specify the word “feel.” So, based on the bubble of warm air around us, what we “feel” is warmer than the actual temperature, and wind chill makes what we feel closer to the actual temperature. So, it could technically “feel” colder than absolute zero, even though that’s a physical impossibility. So the answer to the question asked, would be yes.
I’ve noticed if you rub your hand across a warm frying pan or piece of metal that it feels hotter. Is it a correct assumption that this is similar to the wind chill effect?
That’s exactly right. It’s the opposite of wind chill, but works in exactly the same way.
The answer is “no” due to the word “feel.” Biological entites will freeze solid long before 0K.
Pingback: Why Can’t We Get to Absolute Zero? | MrReid.org
Seems like few people understood the idea of “feel colder than zero”.
We all here know it’s physically impossible, but if we could feel, if the Helium Mr. Physicist said was at 1 K and it was flowing, then it’d feel colder than zero.
my question is -can wind or any movement exist at absolute zero? or could anything be moved or rearranged?