Physicist: Although anti-matter was first experimentally confirmed in 1932, no one has been able to see the atomic spectrum of any anti-elements until December 2016.

The atomic spectra of hydrogen. To see this you get some hydrogen, make it hot, and watch it glow. Easy. Anti-hydrogen (it turns out) has the exact same spectrum, it’s just much more difficult to find out.
The problem with anti-matter is that you can’t let it touch anything. If you do: boom. Although, considering that we can only make it a few atoms at a time, it’s more of a “boom“. Preventing anti-matter from touching things is especially tricky considering how it’s made. Anti-matter doesn’t exist in nature in any great quantities because, like everything else, it’ll eventually bump into something, but unlike everything else, it can only do it once. That means there’s nowhere to find it: you can’t distill or mine anti-matter, you literally have to create it.
We do that by slamming particles together. Whenever there’s enough extra energy in one place new particles are spontaneously generated in particle/anti-particle pairs. Typically, when there’s enough energy around to create new matter and anti-matter, there’s also enough energy to send those new particles sailing.
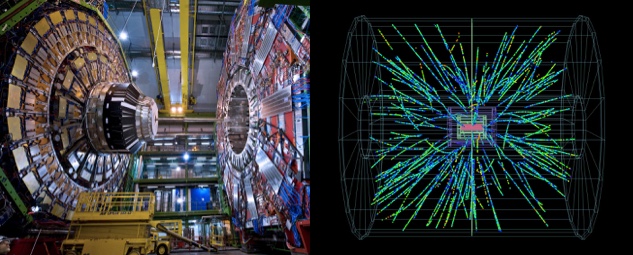
Left: Where lead ions are shot at each other at effectively the speed of light. Right: The tracks of new particles seen by the detectors after a single pair of lead ions hit each other.
After anti-matter is created, you have to slow it down from (nearly) light speed to walking speed and then keep it floating in a hard vacuum using only electromagnetic fields. But electric fields only work on charged particles; neutral matter (which has the same amount of positive and negative charge) isn’t attracted or repelled.
At the same time, atomic spectra is generated by electrons or positrons (anti-electrons) in an atom jumping and dropping between energy levels. But as soon as you bring the anti-protons and positrons together to make anti-hydrogen, you’ve got an electrically neutral atom which promptly falls to the bottom of you container and annihilates with all the force of an ant tapping its foot (individual anti-matter atoms aren’t something to lose sleep over).
Luckily, many atoms, including hydrogen and anti-hydrogen, have a “magnetic moment”. While they are electrically neutral and unresponsive to electric fields, they do act like little bar magnets and we can use that to keep them suspended. Even so, this is no easy task; just for fun, try suspending a magnet in mid-air using other magnets (you’ll quickly discover that failing over and over until you give up is less fun than it is a learning experience).
Not to be deterred by a lack of fun, nuclear physicists cleverly devised a way to keep cold (slow) atoms suspended. The nice, simple rules “opposites attract and likes repel” don’t apply since each atom has both a north and south pole. Instead, we’re forced to rely on a more subtle (weak) effect: hydrogen is “diamagnetic”, meaning that it is repelled by strong magnetic fields.
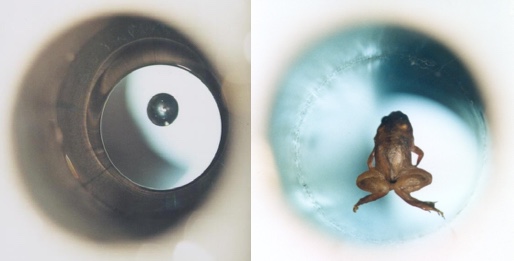
Water, and frogs by extension, are diamagnetic making it possible to float them in the minimum of an absolutely over-the-top strong magnetic field (~16 Tesla). Ironically, this is easier than suspending individual atoms because big collections of atoms that are stuck together (like Kermit here) don’t bounce around nearly as fast.
Even so, combining and suspending fresh-from-the-accelerator anti-protons and positrons in a magnetic trap is akin to catching water from a fire hose in a shallow bowl. At CERN the process of creating anti-hydrogen from anti-protons and positrons is about 28% effective. The process of then catching those atoms is about 0.056% effective. Of the 90,000 anti-protons generated in a given attempt, only an average of 14 graduate to being contained anti-hydrogen.
Once you’re past all of those minor inconveniences, all that remains is to precisely detect the light from a dozen atoms excited by a laser beam. That’s also really difficult, but only because detecting anything about a sample that small is tricky.
The ultimate result, by the way, is that anti-hydrogen has a spectrum indistinguishable from regular, dull-as-dishwater (being a principle component of dishwater) hydrogen. This leaves open the question of why there isn’t more anti-matter in the universe. When we make anti-matter we also make an exactly equal amount of ordinary matter and the same is true of every creation/annihilation process we’re aware of. The expectation is that there’s something fundamentally different about matter and anti-matter that can distinguish between them in a way more profound than “the same, but opposite, y’know?”. What this long awaited experiment shows is that positrons and anti-protons interact with each other in exactly the same way electrons and protons interact (but opposite, y’know?). There are always details and more to explore, so: on to the next thing. For science.







Ah yes, the age old question of ‘why did the universe end up with more matter than antimatter?’
It’s a good question, I’m really looking forward to the day when someone figures out the answer, though I wouldn’t be surprised if I’m long dead by that point.
How do we know that there is more matter than anti-matter? If the spectrum of anti-matter is the same as matter, how do we know that there aren’t whole galaxies out there made of anti-matter?
@Mike Mason
Because galaxies also run into each other sometimes. If even a small fraction of the universe were composed of anti-matter, we’d expect to see huge explosions (of the anti-matter variety) all the time.
There’s also no natural sorting mechanism; neutral matter and anti-matter are effectively identical in every respect, so no one has any good ideas about how you could get all/most of the matter produced (never mind how) to go one way and the anti-matter to go another all while avoiding each other. The situation in the lab is very artificial.
As an anti-matter of fact, I had to re-read this post. Not that I failed to understand it; I was concerned over its proximity to tomorrow’s date.
Please keep up the posts.
Where could be the most of the antimatter in the universe lie about?
Pingback: Q: Why haven’t we been able to see the spectra of anti-hydrogen until recently? Why is it so hard to study anti-matter? | ExtendTree
@Science Enthusiast
High energy interactions produce anti-particles, so you can find them individually flying through space (space is empty enough that an atom can cruise for a long time without hitting anything). Very rarely they can get caught in the magnetic fields of planets and stars, like Earth’s Van Allen Radiation Belt. Even so; it’s on the scale of nanograms.
I’m of the notion that fields preceded space-time; that is, space-time is emergent from the wave activities of interacting/intersecting fields. Fields give us dimensions within space-time by creating space-time. Space-time does NOT give us fields; although particles do spontaneously percolate out of the fields that permeate the Universe suffused with the space-time and its P-wave modulus – which computes to the speed of light in all field frames.
Knots are another fun thing that fields do in wave configurations (knots are mathematically curious in dimensions greater than 3). Knots of fields may exclude space-time and/or loop back upon itself, which may explain the very, very long half-life of some particles, such as a proton.
So on to the topic of anti-particles. Particle spin has chirality. Spin got the Universe started. Perhaps the Universe began construction as fields without space-time to distinguish here from there. The Grand Chirality of the Universe set the stage for the predominance of matter over anti-matter. With local events, such as experimentally-staged, high-energy particle collisions, anti-matter particles will splatter along with ordinary matter. Alas, the Universe does not routinely do head-on high-energy particle collisions to spew out ~50:50 matter:anti-matter. In the scheme of things, the Grand Chirality of the Universe’s spin has favored the survival of ordinary matter…. The End of My Spin