Physicist: The weird effects that show up in quantum mechanics (a lot of them anyway) are due to the wave-nature of the world making itself more apparent. What we normally think of as “particle behavior” is just what happens when the waves you’re talking about are very small (compared to what’s around) and are “decoherent” (which means the frequency, phase, and polarization are all pretty random between one photon and the next). It’s a long way from obvious (there’s some math) but, for example, the way light streams through gaps is governed entirely by the wave-nature of light, and not because it’s a particle.

It looks like the sunlight is made of particles moving in straight lines, but in reality you don’t need “particleness” to describe what’s happening here. The gaps in the door are very large compared to the wavelength of the light (approx. 0.01 m vs. 0.0000005 m). With enough elbow room, waves will proceed in a straight line due to interference effects.
Technically, absolutely everything is fundamentally quantum mechanical. What we consider to be “classical mechanics” is just a special case of quantum mechanics (a large-scale, non-coherent case).
But that’s not at the heart of this question.
Light wears its quantum behavior proudly. The De Broglie wavelength decreases with increasing mass, and while even the lightest particles have fantastically small wavelengths (electrons typically have wavelengths on the order of trillionths of a meter), light can have wavelengths ranging up to miles long (radio waves). Observing quantum effects in matter is difficult, but we see it in light so much that we think it’s normal (which it is, I suppose).
So, other than obvious stuff; like chemistry or… absolutely everything, what follows are some distinctly wavy and quantumy things that you might come across while palling around on this big blue world. Also, this list is far more incomplete than complete, so I’m open to suggestions for things to add.
Technology younger than fifty years old (give or take)
There are a lot of well-established, well-known (in the scientific community) quantum mechanical effects that are in use in almost every fancy device devised since the forties. This include things like lasers (Bose-Einstein statistics), tunnel diodes (quantum tunneling), LED lights, and of course exotic stuff like quantum computers. Unfortunately, the qunatum-ness of modern technology is generally pretty well hidden (and thus: boring).
Using cell phones inside (diffusion)
Ever successfully use a cell phone while inside? Radio waves are huge, so their waveness is very apparent. They can ooze around corners (to some extent), and permeate (non-conductive) materials. It’s a little tricky to describe why radios (including cells phones, wifi signals, etc.) work even when there’s no line-of-sight between the sender and receiver, and especially tricky to describe why they work when there’s no path at all (for example, being in a windowless room with the door closed). I wish I could say it’s quantum tunneling (because that would be awesome), but it’s not quite that. It’s similar to the fact that very long, slow ocean waves will cause your boat to rise and fall as a whole, but very short ocean waves are blocked by things like a boat. Radio waves tend to be about the size of buildings and when they come along, rather than just hitting the walls and bouncing off or getting absorbed, they “raise and lower” the electromagnetic field of the whole area. Already it’s practically impossible to describe radio waves in terms of particles.
Pretty colors (thin-film interference)
There’s an optical device called a “Fabry–Pérot interferometer” which uses wave interference to separate out light of very, very nearly equal frequencies, and it’s basically just two mirrors.
As it happens, thin films of transparent material mimic the F-P interferometer.
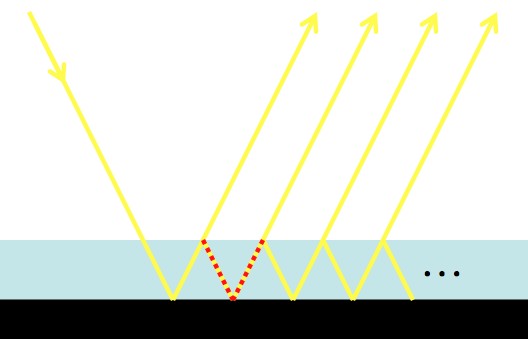
Some light will bounce off of the surface of the film, and some will go through. If the extra length between paths (red dotted line) is a multiple of the light’s wavelength, then you get “constructive interference”. When you get “destructive interference” then there won’t be light reflected at that angle.
Which of the incoming and outgoing angles experiences constructive interference depends on the thickness of the film, the material of the film, and the color of the light. If the light is “monochromatic” (one color or wavelength, like a laser), then this leads to dark areas. If the light has many different colors, then the areas that are dark for some colors may not be dark for others (which leads to prettiness, see the picture below).

Light can bounce back and forth inside of thin, flat, transparent films. Top Left: a green laser and the glass of a mirror. Top Right: sunlight and an oil film on water. Bottom Left: The path diagram for… Bottom Right: “Newton’s rings”.
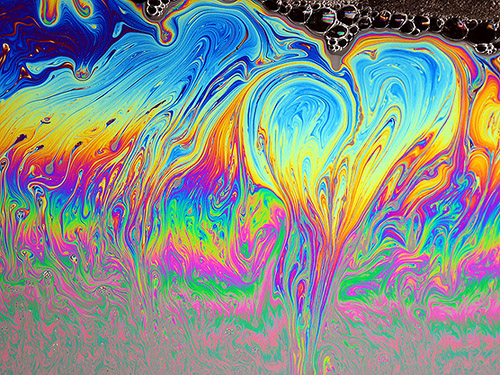
It’s sometimes hard to see, but you can see thin-film effects in soap bubbles as well. In that case the thickness of the film tends to change rapidly, which is why the colors in soap films tend to swirl and change so fast.
True rainbows are formed by another process, but they also ultimately rely on the wave nature of light. It’s just a bit more obscure how.
Dark spots on still lakes (Brewster’s angle and polarized light)
This one is pretty hard to notice. In addition to light’s waviness, it also has polarization (which is a fundamentally not-particle thing to have). The polarization of light affects how it reflects off of a surface (like water) and how it scatters in a gas (like air). If you happen to look at the sky reflecting off of a lake these effects are combined, and at one particular angle they fight each other.
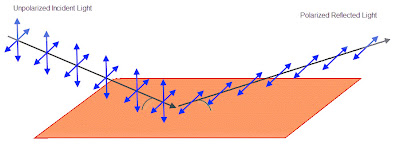
If the angle between the incoming light and the surface of the water is 37°, then only horizontally polarized light will reflect.
The amount of light that reflects off of a surface depends on the polarization of that light, which is why polarized glasses are sold to drivers to cut down on glare. It so happens that if vertically polarized light hits water at about 37° none of it will be reflected (this is called “Brewster’s angle“).
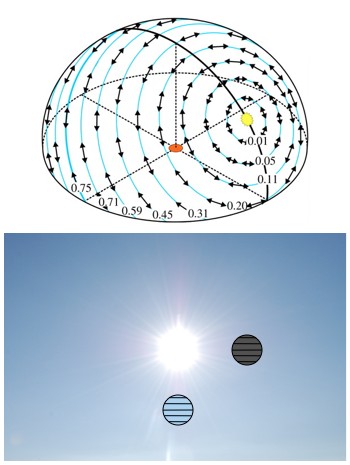
A map of the polarization of the sky (top). The polarization of the blue of the sky “circles the Sun”. So, if you hold up a horizontal polarizer below the Sun it’ll appear clear, but if you hold it up by the side of the Sun it’ll appear dark (bottom).
Because of the way light scatters in air, if you point your hand at any point in the sky (other than the Sun), and turn your palm toward the Sun, then the flat of your hand will be aligned with the polarization of the light coming from that part of the sky. As a result, right around dawn and dusk the entire sky is polarized in the north-south direction.
One consequence of this is that if you’re standing at the right angle early or late in the day, and the sky (not the Sun) is your primary light source, then the face on your digital watch can appear black. Another is that if you look at the sky in a still lake, at about 37° from level, during dawn or dusk, while looking either north or south, you’ll find that the sky isn’t reflected at all and appears black.
For some reason I like this example, in part because it must have been confusing as hell for the occasional fishermen who noticed this over the millennia. However, like the green flash, it’s one of those effects that’s not quite an every-day example.
The door picture is from here, the oil picture is from here, the soap-bubble picture and a description of how it was made can be found here, the reflection picture is from here, and the green-laser-mirror-picture was sent in by a kind-hearted reader as part of a question. The map of the polarizations is from this paper about how bees use that very polarization to navigate. That’s worth pointing out again; many insect species can literally see the polarization of light.







I’m afraid to state “the way light streams through gaps is governed entirely by the wave-nature of light, and not because it’s a particle.” is simply to ignore accepted scientific fact. Observed particles (electrons / photons) leave a pattern of two distinct lines when fired through a double slit – not an interference pattern.
That’s true in part.
Even those two distinct lines are also described entirely by the wave nature of light.
Some other examples:
Birefingence. This aligns nicely with your examples of polarisation. Birefringent crystals (e.g. Calcite) are easy enough to find.
Laser holograms. Not only is interfernce required to produce a holographic film it is also required to regenerate the hologram.
Harlequin paint, carnival glass, the rainbow on a DVD data surface. All get their rainbow colour from interference effects.
Of course, not all quantum mechanical effects are attributed to the wave nature of reality. We have to remember that quantum mechanics also has particle properties. The photo-electric effect, for example, cannot be explained by a wave description of the photon; or perhaps more correctly, it is explained by treating the photon as a wave packet, possessing both a extended (wave) and discrete (particle) structure. The quantisation part of quantum mechanics is equally important.
Great article. Thanks!
I think that superfluid helium is a good example of when quantum mechanical effects become observable, it is frictionless and can do all sorts of strange stuff. I would love if you could explain what is special about superfluid helium, cause I sure can’t!
Isn’t the bulk nature of solids due to the Pauli Exclusion Principle? Solids’ resistance to compression and inability to overlap would not be the same without it. When you sit in a chair and it holds you up without sinking in, thank that quantum effect.
Ok not quite everyday but it’s one of the best things I’ve ever seen, Quantum Locking:
The caption on the photo with the sun and polarizing filters is backwards.
Hi Physicist,
I do not have the training of some of the commentators, but to me the answers are not really addressing the question. Quantum Mechanics appears just a better way of describing what happens, and allows better predictions of what will happen, but you can’t “see” quantum mechanics. Quantum is a way of describing the “particleness” (OK I just made that up) of some forms of energy. the frequency, phase, and polarization of light could equally be features of Wave theory.
I understand Quantum, to be like the Galileo moment. Yesterday, the sun went around the earth, came up over there, went down over there, rose earlier and stayed up longer for a few months, and then rose later and stayed up shorter for a while. The next day we are rushing around the sun. So what changed in every day life, for the ordinary observer, growing food and making things, nothing. What differences could be “seen in everyday life”? Nada.
But we understand that the change of viewpoint altered everything and made current life possible, so with Quantum mechanics.
My (limited) understanding is that Quantum Mechanics is a hypothesis or theory that helps us predict how energy, specifically electro-magnetic energy operates at the very small scale. In every day life we seldom operate at the atomic – photonic level and what we see is “quantum mechanics” at the Macro level.
It helps us design better products, lasers and holographs, we better understand why sometimes in very tight circuits, electrons do not do what we anticipated.
The most important benefit of Quantum mechanics is that it is a necessary step to Quantum Electro Dynamics (QED) and thus is a step towards the next (“the quantum electro everything including gravity, magnetism and why it is there hypothesis”) and an even better understanding of the physics of the worlds around us and within us.
It is easy to create supposes on things that can not be seen. It is very unlikely that the “atomic theory” is true. Look up valence. Even Rutherford the proposer of this suppose doubted it. Neutrons? protons mf Gluons. This stuff is made up unverifiable bs. “There are no Electrons” or gluons. All those chemical equations are right for the wrong reasons. Therefore, wrong. Most of this sophistry “taught” exists to keep active minds busy. It’s proof people can be convinced of anything.
Another example: the unity of consciousness.
The most obvious quantum mechanical effect is the one that got Planck started in the first place: the colors of hot things.
There is no classical explanation of why a moderately hot stove glows dark red and a very hot one glows a brighter, more yellowish color — all the way up to a light bulb filament that glows white hot.
Google for terms like “black body radiation” or “ultraviolet crisis”.
Sorry: “ultraviolet catastrophe” (even a degree in QM can’t stave off some mistakes)
The Brewster’s angle and polarized light is the same phenomenon that causes distant roadway to look “wet” on a bright sunny day
The mirages you might see while driving down the highway are caused by the air near the ground being hotter (and less dense) than the air above it. A solid, general rule of thumb is that waves will turn toward the denser medium, so light from the sky will tilt slightly up.
There is a different effect, that glass and metal reflect polarized light even better than water, that causes some insects to believe that those materials are actually water. But that’s different from a mirage, that’s about insects being easy to fool.
@bob weiss
Beautiful example!
@George LoBuono
There’s a lot of confusion about how quantum mechanics and consciousness are related, and an amazing volume of misleading information has been produced. So far, no evidence has been found for a non-trivial connection between consciousness and quantum mechanics (“non-trivial” here means “more of a connection than a brick has”).
it is the great article , with excellent explanation and illustrations.
Quantum mechanics is useless branch of physics.
quantum mechanics is not useless.if we think of future can we imagine that at the same time we can be on earth as well as on other planet.this is answered in quantum mechanics as debroglie said that there is matter wave .in same way a when a human move than a group of wave is associated with it.in future there can be probablity some wave part can be move to other planet while some remain on earth.quantum mechanics is future science not useless
are there examples of quantum mechanical systems.
Perhaps the most common example of every day quantum at work in a practical, non-classical way is in Flash memory, used in cellphone and many other devices, such as USB “thumb” drives. They depend on tunneling, a quantum phenomenon, to operate.
As noted tunnel diodes do as well, but they are far less common.
Flash memory is the only “everyday” thing I can think of that can NOT be adequately modeled in classical physics. On a practical basis classical physics is usually close enough, so to speak, to describe the everyday world around us. Not that quantum isn’t real. It is, but at human, everyday sort of scale it is “lost in the noise”.
Pingback: Q: What are “actual pictures” of atoms actually pictures of? | Ask a Mathematician / Ask a Physicist
George,
You’re quite right about Flash Memory, but, in fact, no solid-state device can be adequately modeled in classical physics because the very concept of a “semiconductor” relies on the quantum theory of solids. The key parameter of a semiconductor, the bandgap voltage, cannot be predicted or explained using classical techniques.
So, all electronics using transistors or semiconductor diodes rely on quantum mechanics to understand them. That’s why the first thing you learn in a semiconductor physics course is a bit of quantum mechanics!
is its true that we can not predict quantum mechanics to be visible?
HAPPYNESS REWARD,
Yes and no (at the same time).
Carl says “no solid-state device can be adequately modeled in classical physics because the very concept of a “semiconductor” relies on the quantum theory of solids. The key parameter of a semiconductor, the bandgap voltage, cannot be predicted or explained using classical techniques.”
Good point. The potential orbit levels of electrons are quantized, so the way electrons do/don’t jump across band gaps, etc, is quantum. All semiconductors are quantum. Tho when I studied solid-state physics we just “assumed” the electron would or would not jump, based on voltage, etc.
Why do you consider the wave nature of light to be quantum mechanical? It was known well before QM came on the scene. It is the particle nature of light that QM gave us, in particular from studying black body radiation, the photoelectric effect, Compton scattering, e.g. Just because the wave nature of light may be unfamiliar to many, doesn’t make it “quantum mechanical”.
Quantum mechanics is subject to and does not escape from causality
Bruce Barron says: “Quantum mechanics is subject to and does not escape from causality
”
What evidence do you have to support this? There is overwhelming evidence to the contrary.
I apologize for taking so long.
What is a cause? A cause is that which makes a thing to be or makes a thing what it is.
Now the principle of causality states that whatever comes into being must have a cause. This is a self evident principle or axiom and is metaphysically necessary. The principle of causality is an analytic proposition and is universal and necessary. )Neither chance nor probability destroy causality.
The above concerns efficient causality. Thus when I want to make a quantum measurement I reduce the potential quantum state, undetermined, to an actual quantified state using a measuring instrument.
Then there is final causality in which every agent acts for an end. If we are not to explain everything by chance we must admit of finality. This is self evident.
Science has for its own legitimate purposes has reinterpreted causality as predictability. Non predictability replaces causality. But because a thinge is unpredictable does not destroy causality.
There may be some kind of spontaneous self determination within the particle at the last moment, some hidden cause or even an efficient cause not predictable. But there is no violation of any causal or principle
Perhaps you can help me out and demonstrate to me how causality is destroyed or unnecessary.
Pingback: ميكانيكا الكم: هل يمكنن إيجاد أمثلة يومية عنها أم أن رؤيتها لا تتم خارج المختبر؟ - أنا أصدق العلم
I want to study more physical and mathematically concepts from you
Magnets. I’m surprised nobody said magnets.
If I’m not mistaken, isn’t ferromagnetism a quantum effect?